8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(benzoyloxy)-8-methyl-, methyl ester, hydrochloride, 1R-(exo,exo)-.
Methyl 3
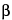
-hydroxy-1
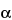 H
H,5
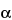 H
H-tropan-2
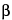
-carboxylate, benzoate (ester) hydrochloride



[
53-21-4].
Packaging and storage—
Preserve in well-closed, light-resistant containers.
Identification—
B:
To 5 mL of a solution (1 in 50) add 5 drops of chromium trioxide solution (1 in 20): a yellow precipitate is formed, and it quickly redissolves when the mixture is shaken gently. Add 1 mL of hydrochloric acid: a permanent, orange-colored crystalline precipitate is formed.
C:
To a solution of about 10 mg in 2 drops of water add 1 mL of 0.1 N potassium permanganate: a violet, crystalline precipitate is formed, and it appears brownish violet when collected on a filter, and shows characteristic, violet-red crystalline aggregates under the low power of a microscope.
D:
It responds to the tests for
Chloride 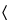 191
191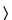
.
Specific rotation 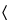 781S
781S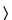 :
:
between
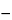
71
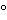
and
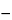
73
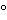
.
Test solution:
20 mg, previously dried, per mL, in water.
Acidity—
Dissolve 500 mg in 10 mL of water, add 1 drop of
methyl red TS, and titrate with 0.020 N sodium hydroxide: not more than 0.50 mL is required to produce a yellow color.
Loss on drying  731
731 —
—
Dry it over silica gel for 3 hours: it loses not more than 1.0% of its weight.
Limit of cinnamyl-cocaine and other reducing substances—
To 5 mL of a solution (1 in 50) add 0.3 mL of 1 N sulfuric acid and 0.10 mL of 0.10 N potassium permanganate: the violet color does not disappear entirely within 30 minutes.
Limit of isoatropyl-cocaine—
Dilute 5 mL of a solution (1 in 50) in a beaker with 80 mL of water, add 0.2 mL of 6 N ammonium hydroxide, stir the solution vigorously during 5 minutes, occasionally rubbing the inner wall of the beaker with a stirring rod: a crystalline precipitate of cocaine is formed, and the supernatant is clear.
Assay—
Dissolve about 500 mg of Cocaine Hydrochloride, accurately weighed, in a mixture of 40 mL of glacial acetic acid and 10 mL of
mercuric acetate TS. Add 2 drops of
quinaldine red TS, and titrate with 0.1 N perchloric acid VS. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 33.98 mg of C
17H
21NO
4·HCl.